‘A very quick onset’
In July of 2018, little Camille came into this world at the St. Paul campus of the Mother Baby Center, a partnership between Allina Health and Children’s Minnesota. In the fall of 2019, and nearing her 15-month check-up, Camille’s parents – Stephanie and Jonathon Ponting – noticed their toddler’s head had started to tilt.
“It was a very quick onset, no warning signs at all,” Stephanie recalled. “Her head was tilting so far that her ear almost touched her shoulder.”
At Camille’s routine checkup, her pediatrician quickly referred her to an eye specialist to start tracking down the cause of the toddler’s head tilt. The doctor determined her eyes were not the reason for the mysterious symptom. The family continued to search for answers with Children’s Minnesota specialists in different clinical areas. During that time, Camille got sick and her parents rushed her to the Emergency Department at Children’s Minnesota in St. Paul. There, the reason for Camille’s head tilt would be revealed.
‘It was surreal’
The Ponting family was anxiously waiting for the results of their daughter’s MRI when they saw new faces walk into their Emergency Department (ED) exam room. They included Dr. Anne Bendel, director of pediatric neuro-oncology at Children’s Minnesota’s cancer and blood disorders program.
“Instantly I was like ‘something is not right,’” Stephanie recalled.
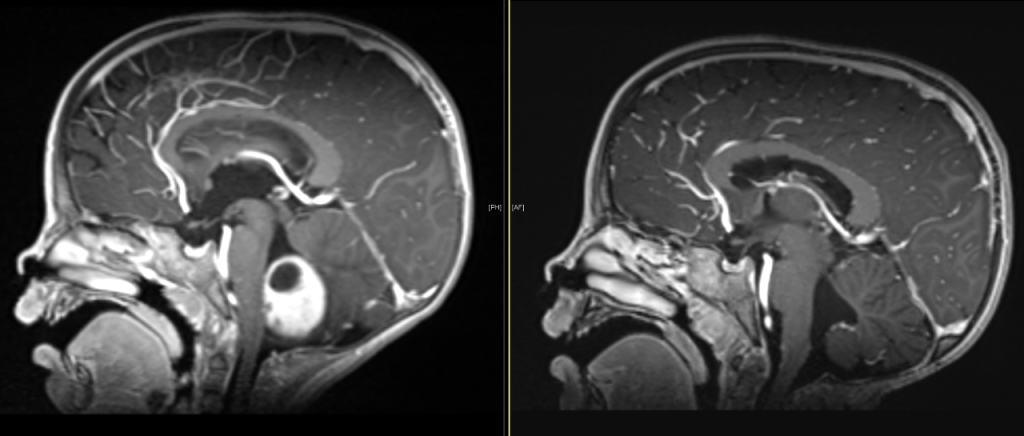
Camille’s MRI scan detected a golf ball-sized tumor in her brain called: pilocytic astrocytoma.
“Pilocytic astrocytoma is the most common childhood brain tumor, accounting for 15% of all brain tumors in children,” explained Dr. Bendel about the potentially deadly diagnosis. “Complete surgical removal of pilocytic astrocytoma gives the best chance of cure, and usually omits the need for any other treatment such as chemotherapy or radiation therapy.”
“I was nervous, scared and confused,” Stephanie recalled. “We learned the tumor was pushing on her brain stem in the area that controls her balance, coordination and movement – that pressure was tilting her head.”
A few days later, Dr. Meysam Kebriaei, medical director of neurosurgery at Children’s Minnesota, met with the family for a surgery consultation. He then became part of Camille’s care team.
“It was surreal, we weren’t expecting this news,” Jonathon described. “The experience in general, and then processing the different people and their titles that we were being introduced to; like ‘Pediatric Neuro-Oncologist’. I can’t say I played a scenario out in my head where this was going to happen. Between Dr. Bendel and Dr. Kebriaei, there was a sense of calmness we received from them.”
Ten days after Camille was rushed to the ED, diagnosed with a brain tumor, and admitted to the hospital – she was being prepped for surgery to remove the tumor. During that time, the Pontings say they formed a bond and trust with Camille’s entire care team.
“Camille would go to Dr. Kebriaei, and he would pick her up and she would refuse to go to anyone else,” Jonathon explained.
“It was like she knew that he was going to fix her,” Stephanie added.

Experimental tumor paint lights the way to recovery
Camille’s family agreed to enroll her in a research trial underway at the time at Children’s Minnesota called, PNOC012: Study of Tozuleristide and the Canvas Imaging System in Pediatric Subjects with CNS Tumors Undergoing Surgery. This trial was a joint effort through the Pacific Pediatric Neuro-oncology Consortium (PNOC), Seattle Children’s Hospital, and Blaze Bioscience through funding provided by Gateway for Cancer Research and the Norcliffe Foundation. The trial specifically studied Tozuleristide, or “tumor paint” to determine if it can help a neurosurgeon better distinguish tumor tissue from normal brain tissue in order to improve the chance of complete tumor removal and lower the risk of injury to the surrounding brain. Before Camille’s procedure, Tozuleristide, a dye-like substance, was injected into her bloodstream. The tumor paint then attached to Camille’s brain tumor tissue and fluoresced under near-infrared light to help Dr. Kebriaei better differentiate between the child’s tumor tissue and normal brain tissue.
“Stephanie and I were looking at it as an extra or best option, the doctors came to us and reassured us,” Jonathon said of his family’s decision to enroll in the trial. “We were like ‘if Dr. Kebriaei feels comfortable with it, and feels it is an extra tool to help him have a successful surgery, then we’re good too.’”
Children’s Minnesota’s cancer and blood disorders research program is a member of several leading organizations, such as PNOC, dedicated to pediatric cancer research and care. PNOC is an international consortium committed to bringing new therapies such as tumor paint to children and young adults with brain tumors. Children’s Minnesota is one of only 25 research sites in the United States, and the only site in Minnesota, to have PNOC trials available for patient enrollment. Camille was one of just 22 Children’s Minnesota patients enrolled in this tumor paint trial.
“Camille’s story is a prime example of what Children’s Minnesota can offer patients through our involvement in experimental therapeutic consortiums,” Dr. Bendel explained. “These collaborative partnerships allow Children’s Minnesota access to different experimental therapies and devices that provide new ways to treat pediatric brain tumors. Our goal with this work is to improve the cure rate and outcome in our patients, and also advance medical knowledge.”
Camille’s brain tumor removal procedure took six-and-a-half hours.
“Dr. Kebriaei told us ‘we got it all, the tumor paint worked and we know we got it all,’” Stephanie recalled. “Her head tilt self-corrected itself almost immediately after the procedure, there have been no side effects and no complications from the procedure.”
“While I’m confident Camille’s procedure would have been a success without the use of Tozuleristide, the tumor paint fluoresced nicely during her procedure. This gave both my team and me, and Camille’s family additional peace of mind,” Dr. Kebriaei said.
The tumor paint trial ran at Children’s Minnesota from 2019 through 2022. As of May 2023, the data from the trial is being analyzed to determine how effective Tozuleristide is at improving outcomes for pediatric brain tumor surgeries.
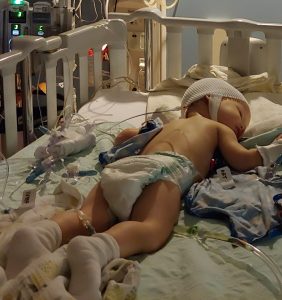
A happy ending on Halloween
On Halloween 2019 – just a week after Camille’s successful brain surgery – she was discharged from the hospital. During her recovery, she was under the watchful eye of the Kid ExpertsTM in Children’s Minnesota’s pediatric intensive care unit (PICU) and neurosurgery patient floor in St. Paul.
“They were all really great,” Stephanie said of Camille’s nurses and care teams.
Camille will still undergo occasional surveillance MRIs. So far, there have been no signs of any regrowth of her brain tumor now two-and-a-half years after her procedure.

Camille’s family shares her story with KSTP
Watch Camille’s story on KSTP.